[Image above] John Goodenough holds a lithium-ion battery cathode material that he invented. Credit: Marsha Miller
John Goodenough has decided that no existent battery is good enough.
Instead, the creator of the cobalt-oxide cathodes that make lithium-ion batteries possible continues to work on the next big thing in the battery world.
Goodenough, a materials science and engineering and mechanical engineering professor at the University of Texas at Austin, and his team of scientists have created another new cathode material—this time to propel sodium-ions into the future power mainstream.
Sodium-ion batteries are promising alternatives to lithium-ions because sodium is much more abundant, and thus cheaper, than lithium. Sodium-ion batteries, however, have been lacking in performance, so scientists are racing to improve these much more affordable power options.
“Although sodium-ion batteries hold tremendous potential, there are obstacles to advancing the technology including issues related to performance, weight, and instability of materials,” according to a press release from the University of Texas.
Goodenough and his team have developed a new cathode for sodium-ions that addresses the batteries’ problem of instability.
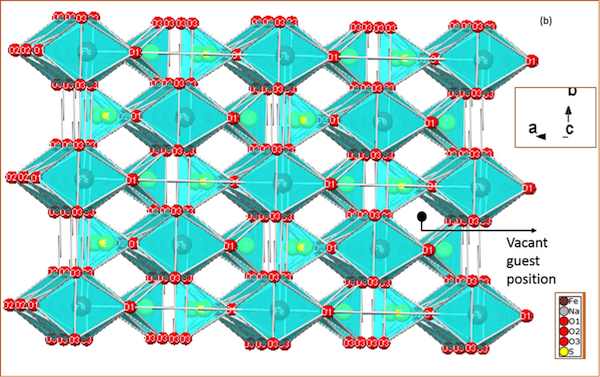
The cathode material is fashioned from the nontoxic and inexpensive mineral eldfellite, NaFe(SO4)2. The mineral, found in volcanic encrustations, is “monoclinic, with yellowish-green, platy crystals.”
The scientists characterized eldfellite and found that the mineral’s crystal sodium and iron layers make for a much more stable cathode material for sodium-ion batteries.
The iron cage provides stability and yet allows sodium ions to flow in and out, a feature essential to create an efficient and useful battery circuit.
Sodium-ions made with the cathode, however, currently have less specific capacity than conventional lithium-ions—two-thirds less, in fact. But that isn’t discouraging its creators.
“There are many more possibilities for this material, and we plan to continue our research,” lead author and researcher Preetam Singh contends in the release. “We believe our cathode material provides a good baseline structure for the development of new materials that could eventually make the sodium-ion battery a commercial reality.”
The paper, published in Energy & Environmental Science, is “Eldfellite, NaFe(SO4)2: an intercalation cathode host for low-cost Na-ion batteries” (DOI: 10.1039/C5EE02274F).