
[Image above] Credit: UCSD
The increased push for fuel cell technologies is putting pressure on the need for more effective solutions for hydrogen fuel storage.
New research from the University of California, San Diego may offer a solution—in the form of our favorite materials. (You said ceramics, not chocolates, right?)
Most fuel cells use hydrogen as fuel. But hydrogen has low energy content by volume, so if you want to use a fuel cell for portable applications (say, to power your car), you’re going to need a substantial volume of hydrogen. Hydrogen also diffuses easily through many materials and is the culprit responsible for hydrogen embrittlement (pdf) of common engineering alloys, including steels, aluminum alloys, and other nonferrous alloys.
According to the US Department of Energy’s Office of Energy Efficiency & Renewable Energy (pdf), an automotive hydrogen fuel cell using current tank storage capabilities would require a hydrogen tank bigger than a standard car trunk to travel just 300 miles. These limitations make current technologies impractical for long-term solutions—so, new advances are needed.
In addition to storage as compressed gas or as liquid in a tank, however, hydrogen can also be stored bound to advanced materials—and ACerS member Olivia Graeve has just the advanced material for the job.
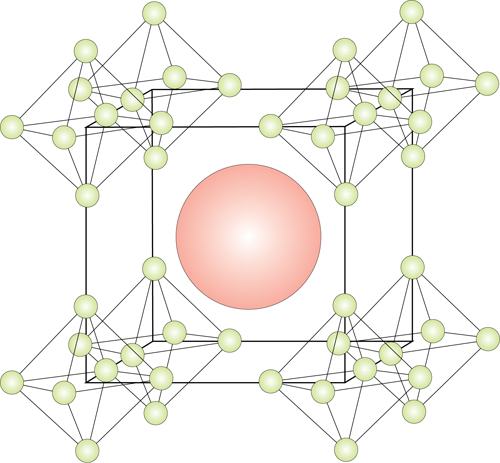
The crystalline structure of new advanced ceramic borides may provide a viable hydrogen storage solution. Credit: UCSD.
Graeve and a team of scientists synthesized compounds of calcium hexaboride, strontium, and various hexaboride mixtures. Using combustion synthesis, they were even able to provide a simple, low-cost production technique for the new compounds.
For combustion, they used a 750°F box furnace to heat boron with metal nitrates and organic fuels, which when ignited, generates heat for the reaction. “It’s a very simple, nice process,” Graeve says in a UCSD press release.
The process synthesized cages of boron molecules containing crystals of the compounds, which can be swapped for hydrogen for an effective storage solution.
According to the press release, “The work is at the proof of concept stage and is part of a $1.2 million project funded by the National Science Foundation, a collaboration between UC San Diego, Alfred University in upstate New York and the University of Nevada, Reno.”
Author
April Gocha
CTT Categories
- Energy
- Material Innovations
- Transportation
Related Posts
Sports-quality ice: From pond side to precision Olympic engineering
February 12, 2026
Solid-state batteries turn heads at CES 2026
January 29, 2026


