
[Image above] Hundreds of thousands of tons of spent batteries are tossed every year, but can ceramics help build better batteries that last longer and require less replacing? Credit: Eva the Weaver; Flickr CC BY-NC-SA 2.0
In today’s world, mobility and flexibility in our gadgets and goods means one thing—batteries. Lots and lots of batteries.
And despite advances to make our mobile power sources last longer and do more, we still tear through packs and pack of batteries annually.
So to achieve a more sustainable, energy-efficient, and environmentally-friendly future, we need better batteries that supply more power and last longer.
Scientists at Oak Ridge National Lab say that ceramics can come to the rescue—they report that testing of a garnet ceramic material suggests it may be just the candidate for tomorrow’s higher-energy batteries.
The most stable batteries realized to date combine a lithium anode, which has the highest known theoretical capacity, with an aqueous electrolyte to shuttle Li ions.
“Many novel batteries adopt these two features, but if you integrate both into a single battery, a problem arises because the water is very reactive when in direct contact with lithium metal,” says lead author Cheng Ma in an ORNL press release. “The reaction is very violent, which is why you need a protective layer around the lithium.”
The materials currently used for that protective layer fall short, so the scientists looked to a material called LLZO—a cubic garnet ceramic composed of lanthanum, lithium, zirconium, and oxygen.
To test how LLZO might fare inside the harsh environment within a battery, the scientists soaked LLZO samples in solutions of varying pH levels ranging from neutral to extremely basic. Then to make sure the material’s structure remained intact, they took an uber up-close look inside using scanning transmission electron microscopy and electron energy loss spectroscopy.
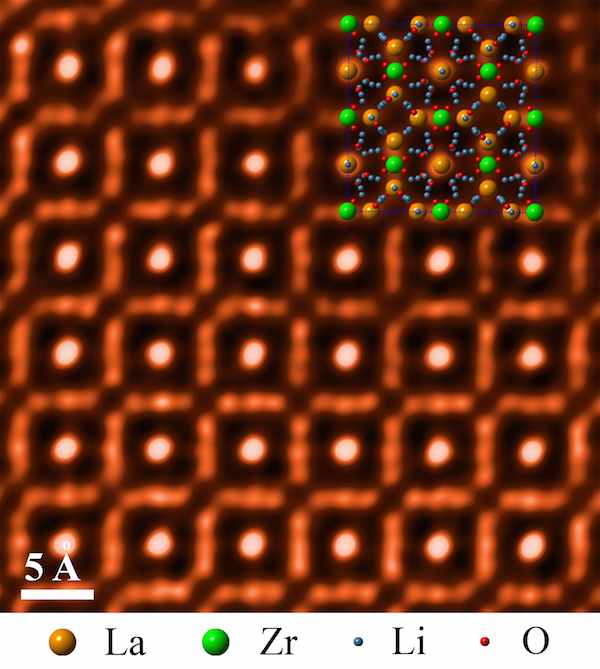
A high-angle annular darkfield scanning transmission electron micrograph of LLZO, with corresponding atomic model overlaid in the upper right. Credit: ORNL
Despite soaking in solutions up to pH 14—as basic as lye and household drain cleaner—LLZO’s structure remained intact, making it a potentially excellent material to keep ‘em separated in better batteries.
“Researchers have searched for a suitable solid electrolyte separator material for years,” says lead author Miaofang Chi in the release. “The requirements for this type of material are very strict. It must be compatible with the lithium anode because lithium is reactive, and it also has to be stable over a wide pH range, because you can have an alkaline environment—especially with lithium air batteries.”
The development of a better protective layer, such as one made up of LLZO, would allow the development of higher-energy density batteries, which can supply more power in a smaller and lighter package—a must for any mobile application.
According to the release, the team is next going to test the material in an operating battery to see how it holds up beyond principle.
The paper, published in Angewandte Chemie, is “Excellent stability of a lithium-ion-conducting solid electrolyte upon reversible Li+/H+ exchange in aqueous solutions” (DOI: 10.1002/anie.201408124).
Author
April Gocha
CTT Categories
- Electronics
- Energy
- Material Innovations


