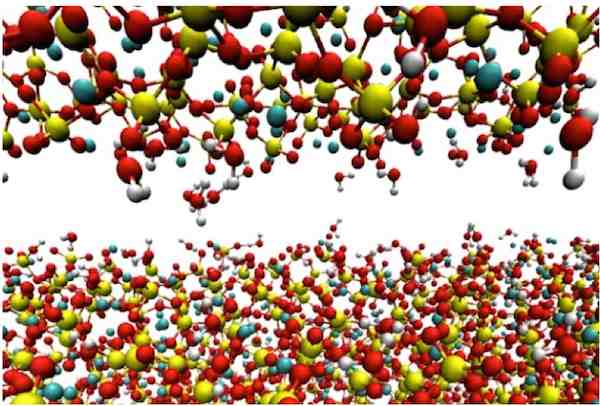
[Image above] A calcium-silicate-hydrate (cement) tip hovers above a smooth tobermorite surface in a computer simulation by Rice University scientists. Credit: Shahsavari Group; Rice University)
Sure, there’s a lot of plastic floating around on this planet. But the most widely used man-made material on earth (by weight)? None other than concrete.
Concrete’s global reign, however, doesn’t mean that it’s the perfect material—lots of researchers are working to improve the ancient material, as evidenced by the amount of times concrete shows up on CTT.
Like our former post about massive supercomputing funds to simulate concrete rheology, new research from ACerS member Rouzbeh Shahsavari, a researcher at Rice University, shows how simulations can help advance the quest for better concrete.
Fellow editor Jessica recently wrote about another of Shahsavari’s research projects, this one about optimizing clinker crushing.
Shahsavari and colleague Saroosh Jalilvand computationally examined the interactions of atoms within a structurally complex material, using concrete as an example, to see how atomic interactions affect the material’s mechanical properties. In addition to concrete, the team’s work could help improve other noncrystalline materials, such as ceramics, sands, powders, grains, and colloids, according to a Rice University press release.
Concrete is a garbled mix of particles with a complex microstructure. What’s inside? Mostly water, aggregate (larger chunks of rock, material, and such), and cement. Cement—calcium-silicate-hydrate (C-S-H)—acts as the glue that holds it all together. Previous research by MIT scientists revealed that cement is not quite crystalline and not quite amorphous—it’s ordered somewhere in between.
[Lots more cement-based research and advances can be found at the 6th Advances in Cement-based Materials meeting, July 20–22, in Manhattan, Kan. Abstracts are due by March 31!]
While heterogeneity makes the material strong, it also makes it hard to predict how interactions within the material affect its overall properties. According to the Shahsavari, the forces between the atoms are critical for the concrete’s overall strength and fracture properties.
“Understanding interparticle interactions is of paramount importance when it comes to mechanics of particulate materials such as cementitious materials or ceramics,” Shahsavari explains in an email. “This work, for the first time, put an atomistic ‘lens’ to decode the interplay between chemistry and mechanics for complex interfacial interactions of cementitious materials. As such, the work introduces exciting new opportunities to better understand the true origin of ‘friction’ and ‘contact’ in these materials and thus be better positioned to tune the mechanical properties of particulate systems. For instance, by identifying the relative importance and quantitative contribution of each atom type to the frictional properties, our works suggest that new processing routes are required to put the right elements at the interfaces of the particles, rather than putting them inside the particles or randomly distributed in the mix.”
To get a better sense of what is going on within the material, the researchers created a computational model of concrete’s microstructure, modeling both rough C-S-H and smooth tobermorite (a calcium silicate hydrate mineral).
In this virtual world, the researchers dragged a virtual tip of C-S-H across a tobermorite surface, measuring the correlation between the force of the push and the corresponding displacement of atoms. Watch the video below to see this simulation in action.

Credit: NS Rice CEVE; YouTube
The simulations correctly predicted the fracture toughness of tobermorite, which has been previously measured—indicating to the researchers that they were on the right path—and helped provide insight into cementitious mechanics.
The press release elaborates:
“Classical studies of friction on materials have been around for centuries,” [Shahsavari] said. “It is known that if you make a surface rough, friction is going to increase. That’s a common technique in industry to prevent sliding: Rough surfaces block each other.
“What we discovered is that, besides those common mechanical roughening techniques, modulation of surface chemistry, which is less intuitive, can significantly affect the friction and thus the mechanical properties of the particulate system.”
Shahsavari said it’s a misconception that the bulk amount of a single element—for example, calcium in C-S-H—directly controls the mechanical properties of a particulate system. “We found that what controls properties inside particles could be completely different from what controls their surface interactions,” he said. While more calcium content at the surface would improve friction and thus the strength of the assembly, lower calcium content would benefit the strength of individual particles.
“This may seem contradictory, but it suggests that to achieve optimum mechanical properties for a particle system, new synthetic and processing conditions must be devised to place the elements in the right places,” he said.
The researchers also found the contribution of natural van der Waals attraction between molecules to be far more significant than Coulombic (electrostatic) forces in C-S-H. That, too, was primarily due to calcium, Shahsavari said.

Computer simulation of a calcium-silicate-hydrate (cement) tip sliding across a smooth tobermorite surface. Credit: Shahsavari Group
The researchers think their work will help illuminate new ways to adjust the chemistry of cement to make it stronger and to tailor it for specific applications.
The paper, published in ACS Applied Materials & Interfaces, is “Molecular mechanistic origin of nanoscale contact, friction, and scratch in complex particulate systems” (DOI: 10.1021/am506411h).

A top-down look at the tip of a virtual cement probe shows the position of atoms, dominated by calcium and silicate. Credit: Shahsavari Group; Rice University
Author
April Gocha
CTT Categories
- Basic Science
- Cement
- Construction
- Modeling & Simulation



