[Image above] Transparent nanoceramics exhibit material properties, such as optical transmittance, that exceed current transparent ceramics containing micro-sized grains. Finding an easy way to synthesize transparent nanoceramics, though, is challenging. Credit: Wen et al., Advanced Science (CC BY 4.0)
As our Wednesday video last week showed, transparent ceramics are garnering interest across a wide range of applications, including lasers, armor, and infrared domes. Compared to conventional glass and single-crystal technologies, transparent ceramics offer similar transparency but are stronger and harder than glass and can be shaped more freely than single crystals.
Transparent ceramics are clear because their polycrystalline microstructure is made of submicrometer, intimately stacked crystalline grains that allow light to propagate with minimal scattering.
But researchers are interested in continuing to downsize the grains to the nanoscale (1,000-times smaller) because nanoceramics are expected to exhibit material properties that could exceed current transparent ceramics.
Creating transparent nanoceramics, though, is challenging. Many currently used synthesis methods are based on powder sintering and, in addition to requiring strict control over experimental conditions, are accompanied by undesired rapid grain growth. More extreme synthesis techniques, including super‐high pressure and spark plasma sintering, may shift grain size back toward the submicrometer range.
Many scientists are investigating new synthesis strategies to produce quality transparent nanoceramics, and in today’s CTT, we take a look at two studies investigating one promising strategy—pressureless bulk glass crystallization.
What is bulk glass crystallization?
Bulk glass crystallization is a promising alternative strategy to powder sintering synthesis of nanoceramics that could make it easier to create large-scale, shapeable transparent ceramics.
In bulk glass crystallization, a bulk glass precursor is subjected to an appropriate heat treatment to cause the bulk glass to crystallize. Scientists have used bulk glass crystallization to create transparent ceramics, but no one had used the method to create transparent nanoceramics before, according to a Nature Communications open-access paper from 2018.
Pressureless crystallization of transparent nanoceramics
The 2018 study on pressureless glass crystallization was conducted by researchers from various institutions in China and France. In the introduction to their study, they claim that “… pressureless fabrication of transparent nanoceramics has never been reported up to date.”
When the researchers say “pressureless,” they do not mean the process takes place in a vacuum. Rather, a pressureless fabrication method simply means there is no applied pressure—fabrication takes place at room pressure.
The researchers tried to create transparent yttrium aluminum garnet (YAG)-based crystalline materials via complete nanocrystallization of a 74 mol% Al2O3–26 mol% Y2O3 (AY26) bulk glass. The transparent bulk glass precursor was synthesized using an aerodynamic levitation system equipped with a CO2 laser, and then the bulk glass was put through a single thermal treatment of 2 hours at temperatures of 950º–1,100°C to induce crystallization.
The researchers found that the size of both YAG and γ-Al2O3 nanocrystals were quite homogeneous and that the nanocrystals could be tailored as a function of the temperature and duration of the single crystallization heat treatment.
Compared to YAG single-crystal and traditional transparent ceramics, the YAG-based nanoceramic showed comparable or superior mechanical, optical, and thermal properties. As the researchers conclude, “these YAG-Al2O3 ceramics are believed to be promising candidates for wide optical applications such as gem stones, lenses, scintillators and phosphor converters.”

HRTEM micrograph of the YAG-Al2O3 ceramic showing the presence of two crystalline phases: thin Al2O3 areas surrounding YAG nanograins sharing grain boundaries (scale bar corresponds to 20 nm). This microstructure is typical of a coalescence growth mechanism. Credit: Ma et al., Nature Communications (CC BY 4.0)
The open-access paper, published in Nature Communications, is “Pressureless glass crystallization of transparent yttrium aluminum garnet-based nanoceramics” (DOI: 10.1038/s41467-018-03467-7).
Pressureless crystallization of nanoceramic fiber
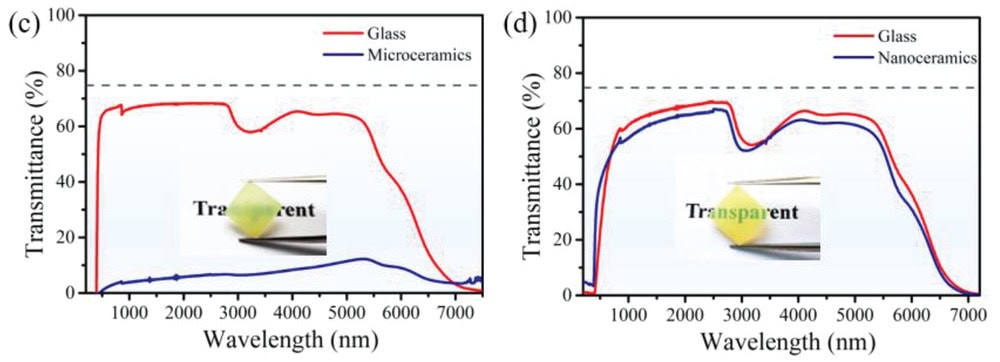
In an open-access study published this June in Advanced Science, researchers from China also created transparent nanoceramics through pressureless glass crystallization. But instead of using bulk glass as the precursor, they used a glass rod instead.
“The nanocrystallization of fiber was achieved by the same [heat]-treatment procedure similar to bulk nanoceramics,” the researchers say in the paper.
To create the precursor glass rod, the researchers melt-quenched a typical tellurite multicomponent glass and then poured the melt into a mold, pressing it with a brass plate to form the parent glass. They obtained the transparent crystallized nanoceramic fibers after heat-treating the glass rod at 440°C for 1.5 hours.
The nanoceramic fibers exhibited excellent mechanical and optical properties, specifically higher hardness (466 HV) than microceramics (438 HV). The researchers also noted the nanoceramics showed an increase in crystalline volume fraction, which “may lead to more compact microstructure [and] that is highly favorable for further improving the mechanical properties.”
According to the researchers, the novelty of their method is embodied in the creation of a sharp contrast between nucleation and growth tendency.
“By intentional creation of a sharp contrast between nucleation and growth rates, the crystal growth rate during glass crystallization can be significantly suppressed,” they say in the paper. “Importantly, this unique phase‐transition habit enables the achievement of transparent nanoceramics and even smooth fibers with extremely tiny crystalline size (≈20 nm) and high crystallinity (≈97%) under atmospheric pressure.”

A schematic illustrating the strategy for steering crystallization habit of glass. a) The classical curves of temperature-dependent nucleation and growth rate. b,c) Reshaping the crystallization rate curves via suppression or enhancement of nucleation rate leads to (b) microcrystallization or (c) nanocrystallization, respectively. Credit: Wen et al., Advanced Science (CC BY 4.0)
The open-access paper, published in Advanced Science, is “Pressureless crystallization of glass for transparent nanoceramics” (DOI: 10.1002/advs.201901096).
Author
Lisa McDonald
CTT Categories
- Glass
- Nanomaterials
- Optics
Related Posts
Progress continues in 3D printing of glass
March 19, 2026


