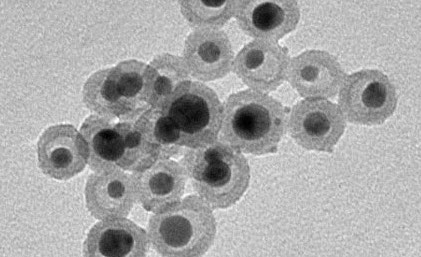
Silver nanoparticles encased in silica shells resemble little rattles—nanorattles—and may provide safe antimicrobial coatings for biomedical implants. Credit: Swiss National Science Foundation.
Silver is a great antimicrobial agent—the metal’s ions permeabilize bacterial membranes and interrupt bacterial metabolism, but are relatively (although debately) safe to human cells.
However, high local concentrations of silver ions are undesirable (see the effects of permanent silver deposition in the skin, called argyria). Therefore, the ability to slowly release silver nanoparticles in biomedical settings would be optimal for safe prevention of biofilm growth on biomedical implants. Biofilm growth on implants is a big problem after surgery, usually requiring additional surgery to remove the biofilm-laden implant and replace it with a clean one.
Scientists from the University of Fribourg (Switzerland), led by Katharina Fromm, may have figured out a way to improve biomedical implants to prevent biofilm growth. The work, published in the upcoming issue of Particle & Particle Systems Characterization, details the synthesis of silica-encased silver nanoparticles in a single-pot microemulsion recipe. Their method formed uniform silver nanoparticles within reverse micelles in an oil-water emulsion. Then, by adding silica precursors aminopropyl-trimethoxysilane (APTS) and tetraethyl orthosilicate (TEOS), they synthesized silica capsules around the silver nanoparticle-containing micelles.
The process resulted in a uniform mix of silver nanoparticles (4-10 nm in size) encased in hollow porous silica shells that were 13-20 nm in diameter. Under an electron microscope, the hollow void between the silver and the silica made the particles resemble little rattles—nanorattles.
The team found that they could easily manipulate the qualities of the nanorattles, providing a simple means to tailor them for a specific application. The scientists showed that increasing the concentration of silver ions in the starting solution could increase the size of the nanoparticles and their concentration within the shells, adjust the shape of the silver nanoparticles, or adjust the size of the void. They could also alter the size of the silver nanoparticles and the thickness of the silica shell by adjusting the concentrations of additional chemicals in the starting reaction.
Further in vitro experiments confirmed that the silver nanoparticles slowly released from the nanorattles through pores in the silica shells. The team is now working on in vivo testing of the nanorattles and is collaborating with another UF group to engineer them for “targeted release,” according to a press release from the Swiss National Science Foundation, which helped fund the research. To achieve targeted release, the team is exploring how to attach bacterial sensors to the silica shells such that silver nanoparticles would release only in the presence of bacteria, thereby reducing patients’ silver exposure.
In addition, the press release states that the nanorattles could also have potential applications as nanoreactors for chemical reactions and for the development of novel batteries.
“These nanorattles have many advantages,” senior author Fromm said in an email. “For the biomedical aspect, they are better than naked silver nanoparticles, because the cells seem to tolerate much better the ‘wrapped up’ silver as compared to the naked silver. In terms of catalysis, one can profit from all kinds of silver-based catalysis, but can also limit access to nanoparticles by tuning the shell thickness and porosity.”
The article is “One-Pot Synthesis and Catalytic Properties of Encapsulated Silver Nanoparticles in Silica Nanocontainers” (DOI: 10.1002/ppsc.201300304).
