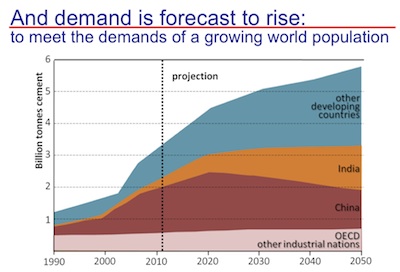
World projected concrete demand. Credit: Karen Scrivener.
Nature offers a new, free-access story by Ivan Amato that updates some of the efforts to lower CO2 emissions and the energy requirements for cement production going on at the Concrete Sustainability Hub (CSHub) at the Massachusetts Institute of Technology in Cambridge. The article particularly focuses on their work to make progress in modeling the calcium silicate hydrate (C-S-H) reactions, and understanding the difference characteristics of its alite and belite phases.
While the story, “Green Cement: Concrete Solution” covers some fairly familiar (if not somewhat misleading) grounds, it does suggest that CSHub researchers are getting a better handle on the pros and cons of increasing the belite content of clinker. Allow me to make a few verbal shortcuts here, but, in brief, clinker is eventually ground into a powder that is known as Portland cement. The alite phase, abbreviated in cement nomenclature as C3S, is more reactive with water and therefore hydrates (cures) in concrete mixtures faster than belite. The downside to alite is that it requires more energy and CO2 emissions than belite (alite forming about 1,500°C and belite around 1,200°C. Thus, the thinking goes that if there was a clever way to make better use of belite, there would be a payoff to the environment.
One group of researchers at the CSHub report in a recent newsletter (pdf) that they have been investigating the various polymorphs of belite, specifically its Beta β), Gamma (γ), and three Alpha (α) phases. In particular, they have been using “first-principles fully quantum mechanical analytical techniques” to model the different polymorphs. They have combined the modeling with statistical analyses to focus on and predict the most reactive and favorable structures of β and γ, and claim to have a deeper understand of how adatoms (atoms adsorbed into the crystal structure) and defects play a role in determining surface energies and crystal reactivity.
As Amato correctly notes, there is another downside to belite, besides lower reactivity: it is harder and therefore requires more mechanical energy to grind, a reality that decreases any potential net energy savings.
Some experts, such as Karen Scrivener who leads a European-based research initiative (and who I interviewed last year), have doubts that there will be a large payoff to tinkering with the clinker composition. Instead, Scrivener and others think larger gains can be made my replacing a significant amount of the clinker (“reducing the clinker factor”) by using supplementary cementitious materials (SCMs) that, conveniently, are often byproducts of other industries. These SCMs can include fine limestone, flash, blast furnace slag, silica flume, and natural pozzolans. In fact, the US may lag Europe and other regions in the use of SCMs, which Scrivener says European cement producers over the last two decades have successfully found substitutes for about 25 percent of the clinker.
One major difference, however, between Europe and the US is that the concrete industry, in the former, tends to use mixes containing SCMs that are pre-made at cement-making facilities, while US concrete makers who do use SCMs tend to add the SCMs while mixing the concrete. Waiting until the concrete-mixing stage introduces a significant change for variations from batch to batch.
Amato does reports on efforts by at least one US company, Ceratech, to produce and market cement mixes that contain flash. But Amato notes, “Ceratech is a bit player in the cement industry, and its approach to cutting carbon emissions amounts to a mere clink in a multibillion-tonne batch. The big carbon reductions will come only when next-generation cements are embraced by the construction industry’s thousands of independent producers, engineers, architects, city planners, and building inspectors.”
But above I mentioned that I thought there are some misleading points to Amato’s article, starting with the title (which, in fairness he may not have written). The admittedly nonintuitive truth is that concrete is already a relatively “green” construction material that is far less energy intensive than masonry, steel, aluminum, and even wood. Another inconvenient truth is that cement and concrete construction are absolutely essential to housing and infrastructure development, especially in the developing regions, and that it is a pipe dream to imagine some alternative in the remainder of the 21st Century. A final inconvenient truth is that the availability of some of the SCMs is actually very small—including waste slag and fly ash—compared to overall production quantities—and in some cases nonexistent because the regions lack the steelmills and powerplants that generate the byproduct. Therefore, research into the use of more common SCM sources, such as calcined clays and natural pozzolans is expected yield the most significant sustainability breakthroughs in the future. Scrivener says that a 10 percent reduction in the amount of CO2 currently associated with the production of cubic meter of concrete would be equivalent to removing all of the CO2 emissions associated with steel production.
All of this is not to say that CSHub’s work is not significant. In fact, it appears that they are doing a lot of valuable computation modeling work that will contribute to worldwide efforts to understand hydration and to build databases that can be used to create custom cement and concrete mixes based on regionally available sustainable resources. Even one or two percent efficiency gains in clinker production are of value given the enormous amount of cement and concrete produced world wide.
I would be remiss if I didn’t note that 4th Advances in Cement-Based Materials meeting, coorganized by Cements Division of ACerS and the Center for Advanced Cement-based Materials, will be held July 8-10 at University of Illinois at Urbana-Champaign. (Abstract deadline is March 25).
